
Document Lifecycle Management
for Regulated Suppliers
Why Uncontrolled Document Lifecycles
Fail Audits
ValidaPoint configures Microsoft 365 to enforce structured lifecycle management across your document control system. All data remains inside your own domain. No additional software licences required.
100%
100%
Validated System Control
Validated System Control
100%
100%
Data sovereignty retained
Data sovereignty retained
0
0
Obsolete document findings
Obsolete document findings
Why Uncontrolled
Document Lifecycles Fail Regulatory Audits
Why Uncontrolled Document Lifecycles
Fail Audits
Uncontrolled document lifecycle management produces the most common findings across regulated industries. These findings signal a loss of system control to external inspectors.
No Enforced Lifecycle Stages
Without formal lifecycle governance, personnel cannot distinguish a draft from an approved version or a retired procedure. Obsolete files remain in active use across operational areas. Auditors identify them as major non-conformances.
Without formal lifecycle governance, personnel cannot distinguish a draft from an approved version or a retired procedure. Obsolete files remain in active use across operational areas. Auditors identify them as major non-conformances.
Approval Processes Without
Traceability
Routing documents through email for review produces approvals that cannot withstand Part 11 scrutiny. No immutable record exists of who reviewed, when approval occurred, or under which version stakeholders accepted responsibility.
Routing documents through email for review produces approvals that cannot withstand Part 11 scrutiny. No immutable record exists of who reviewed, when approval occurred, or under which version stakeholders accepted responsibility.
Lifecycle Failures Carry
Commercial Consequences
Poor information management triggers formal observations during customer audits. Each finding demands a documented CAPA response, exposes the organisation to Warning Letters, and places approved supplier status under review.
Poor information management triggers formal observations during customer audits. Each finding demands a documented CAPA response, exposes the organisation to Warning Letters, and places approved supplier status under review.
Controlled. Traceable. Audit-Defensible.
ValidaPoint configures Microsoft 365 to enforce every lifecycle stage automatically, from creation through final disposal. All documents, metadata, and audit trails remain within your own domain.

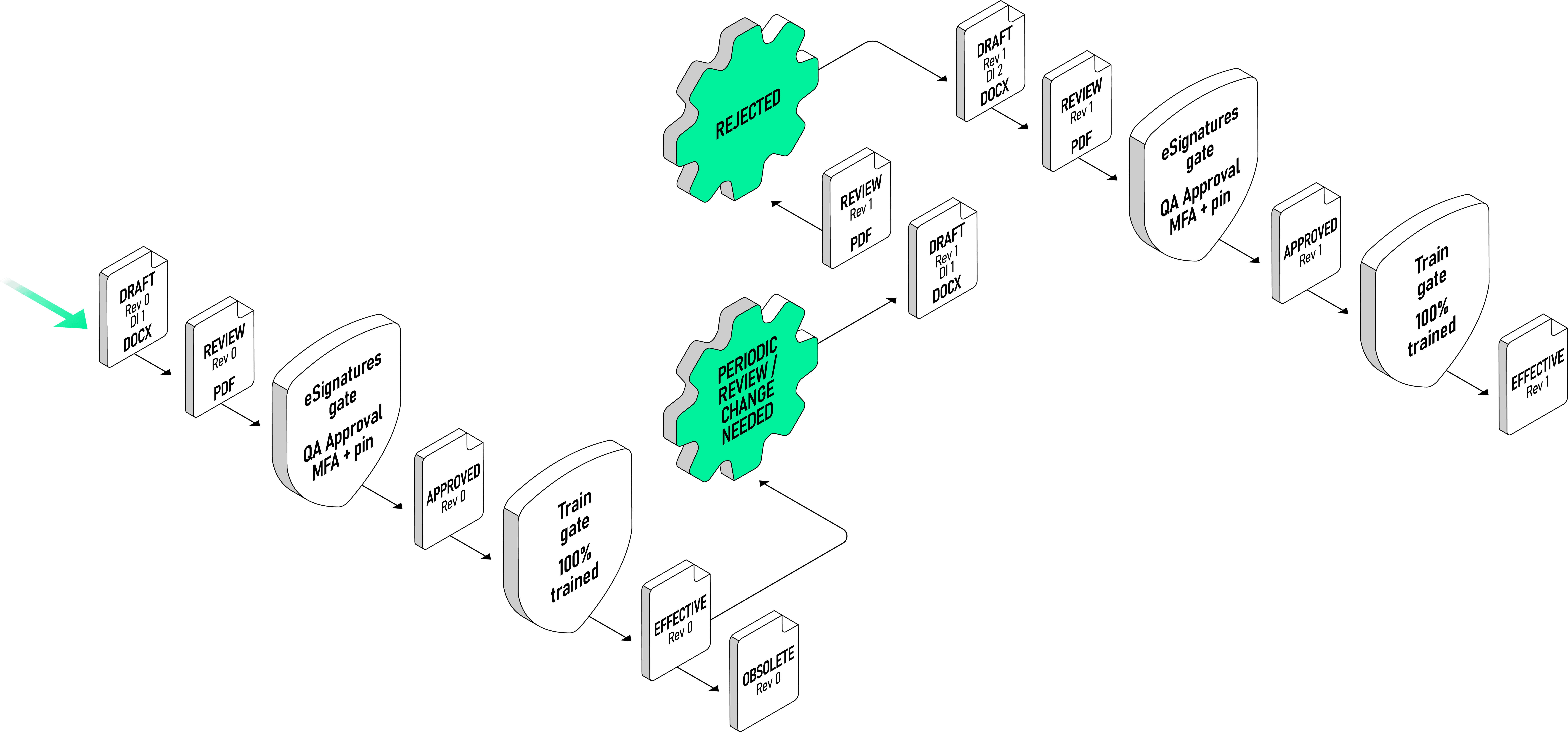
Enforced Lifecycle State Transitions
Strict status controls govern how documents move between stages. No document progresses without satisfying defined conditions. Unauthorised edits, premature distribution, and informal bypasses are blocked at the system level.

Enforces: Draft → Under Review → Approved → Effective → Obsolete

Prevents progression without required approvals

Retires superseded versions automatically upon new effective date

Blocks access to documents in active development for unauthorised users
Structured Review and Approval Workflows
Role-based routing delivers documents to authorised reviewers automatically. Cryptographic electronic signatures permanently link identity, role, and timestamp to a specific version. The configuration satisfies FDA 21 CFR Part 11 and EU GMP Annex 11 requirements.

Routes documents to designated stakeholders without manual effort

Captures role, identity, timestamp, and version at point of signature

Supports structured collaboration across departments

Exports complete approval chain evidence for retrieval during audits


Controlled Distribution and Secure Access
Distribution follows defined permission structures once a document reaches effective status. Authorised users access only current approved versions. Every retrieval, download, and print action is logged in an immutable audit trail.

Restricts access to authorised users by role and department

Logs every retrieval, download, and print with user identity and timestamp

Prevents obsolete documents from reaching operational areas

Governs document sharing with external partners within controlled access boundaries
Every Lifecycle Stage Is Fully Governed
ValidaPoint enforces the complete lifecycle within Microsoft 365, from creation through destruction.
Creation
Creation
New documents enter the system within defined templates. Mandatory metadata is assigned at creation: version, owner, department, and status. No uncontrolled file enters active workflows.
New documents enter the system within defined templates. Mandatory metadata is assigned at creation: version, owner, department, and status. No uncontrolled file enters active workflows.
Review and Approval
Review and Approval
Structured workflows route documents to the correct stakeholders. Cryptographic signatures capture the full approval record before any document reaches active use.
Structured workflows route documents to the correct stakeholders. Cryptographic signatures capture the full approval record before any document reaches active use.
Active Use and Retention
Active Use and Retention
Access controls enforce that only current effective versions are retrievable. Retention practices maintain records for the required product lifecycle and support ongoing compliance reviews without manual effort.
Access controls enforce that only current effective versions are retrievable. Retention practices maintain records for the required product lifecycle and support ongoing compliance reviews without manual effort.
Archiving and Final Disposal
Archiving and Final Disposal
Upon retirement, documents are securely archived with full history intact. Final disposal follows defined policy requirements. Destruction records are retained as objective evidence of formal lifecycle closure.
Upon retirement, documents are securely archived with full history intact. Final disposal follows defined policy requirements. Destruction records are retained as objective evidence of formal lifecycle closure.
Benchmark Document Management Against Audit Expectations
Identify gaps in version control, approval traceability, and access management before your next customer audit.
A short assessment to benchmark your current practices
against audit-ready expectations.
Protecting Lifecycle Integrity Across Your Quality System
The validated architecture protects data integrity and operational efficiency across every compliance scenario.
Single Source of Truth

Enforces one authoritative version at every lifecycle stage

Authorised users access only the current effective document at point of use

Prior versions securely archived and retrieved during compliance reviews without exposure to active workflows

Eliminates version conflicts as a root cause of obsolete document findings

Protecting Lifecycle Integrity Across Your Quality System
The validated architecture protects data integrity and operational efficiency across every compliance scenario.
Single Source of Truth

Enforces one authoritative version at every lifecycle stage

Authorised users access only the current effective document at point of use

Prior versions securely archived and retrieved during compliance reviews without exposure to active workflows

Eliminates version conflicts as a root cause of obsolete document findings

Protecting Lifecycle Integrity Across Your Quality System
The validated architecture protects data integrity and operational efficiency across every compliance scenario.
Single Source of Truth

Enforces one authoritative version at every lifecycle stage

Authorised users access only the current effective document at point of use

Prior versions securely archived and retrieved during compliance reviews without exposure to active workflows

Eliminates version conflicts as a root cause of obsolete document findings

Aligned with
Global Regulatory Standards
The ValidaPoint configuration aligns with the regulatory standards governing document lifecycle management across the life sciences supply chain.
FDA 21 CFR Part 11
Part 11 requires computer-generated, time-stamped records governing document creation, review, approval, and distribution. ValidaPoint enforces lifecycle controls and generates tamper-proof records satisfying these requirements. All records remain within the organisation's Microsoft 365 tenant.
EU GMP Annex 11
ISO Standards (ISO 9001, ISO 13485, ISO 15378)
GDP and GMP Supply Chain Requirements
Replacing Manual Operations
with Structured Control
Structured lifecycle management eliminates the compliance gaps that drive recurring audit observations.
Feature
Feature
Lifecycle State Enforcement
Lifecycle State Enforcement
Approval Traceability
Approval Traceability
Obsolete Document Control
Obsolete Document Control
Metadata Governance
Metadata Governance
Change Management
Change Management
Audit Evidence Retrieval
Audit Evidence Retrieval
Manual and
Hybrid Systems
Manual and
Hybrid Systems
No formal stages. Documents freely modified at any point.
No formal stages. Documents freely modified at any point.
Email threads with no compliant audit record
Email threads with no compliant audit record
Outdated versions remain accessible to all users
Outdated versions remain accessible to all users
Inconsistent, manually maintained across files
Inconsistent, manually maintained across files
Informal, undocumented revisions with no impact assessment
Informal, undocumented revisions with no impact assessment
Manual reconstruction under time pressure
Manual reconstruction under time pressure
ValidaPoint
Configuration
ValidaPoint
Configuration
Strict Draft → Approved → Effective → Obsolete controls
Strict Draft → Approved → Effective → Obsolete controls
Cryptographic signatures linked to version, role, and timestamp
Cryptographic signatures linked to version, role, and timestamp
Automatic retirement upon new effective date
Automatic retirement upon new effective date
Mandatory fields enforced at creation. No exceptions.
Mandatory fields enforced at creation. No exceptions.
Controlled versioning with full change history and audit trail
Controlled versioning with full change history and audit trail
Structured records retrieved on demand
Structured records retrieved on demand
Validated Implementation: Structured Deployment for Audit Defensibility
Validated Implementation: Structured Deployment for Audit Defensibility
Implementation follows a QA-approved validation methodology without disrupting ongoing supply chain operations.
Implementation follows a QA-approved validation methodology without disrupting ongoing supply chain operations.
Phase 1: Discovery and Scope
Requirements gathering, document lifecycle gap analysis, and validation scope definition aligned to your organisation's regulatory requirements.
Phase 2: Configuration
Azure deployment, document library structuring, lifecycle state configuration, and automated approval workflow setup within your Microsoft 365 tenant.
Phase 3: System Validation
Execution of pre-built IQ/OQ/PQ test scripts against lifecycle workflows. QA-ready validation reports generated as evidence of intended use.
Phase 4: Production Go-Live
Targeted end-user training and full production deployment. Users across operational and quality teams manage documents in a validated state of control from day one.



Eliminate Lifecycle Gaps
Before Your Next Audit
Benchmark current document management practices against GxP auditor expectations. Identify version control vulnerabilities,
approval traceability gaps, and distribution control risks before a customer does.
Diagnostic criteria

Lifecycle state enforcement reliability

Approval and Part 11 electronic signature traceability

Obsolete document access controls

Metadata completeness and integrity

Training linkage to effective document versions
Our Other Capabilities
Built on:
Built on:
Audit-Ready Document Control for Microsoft 365
ValidaPoint configures Microsoft 365 as a GxP-compliant document control system.
ValidaPoint runs on infrastructure certified with ISO 27001 (Microsoft Azure), ISO 9001 (Microsoft Azure) and SOC 2 Type II (Microsoft Azure)
